Medical Consumables interventional high pressure balloon Catheter (HP PTA)
INTENDED USE
The Ultra WANTY HP PTA Balloon Catheters are indicated for peripheral transluminal angioplasty (PTA) for peripheral vascular system (including the iliac artery, femoral artery, iliac femoral artery, popliteal artery and inferior genicular artery).
Description of high pressure balloon Catheter
The Ultra WANTY high pressure balloon Catheter is an over the wire (OTW) peripheral balloon catheter, specifically designed for percutaneous transluminal angioplasty in atherosclerotic obstructed vessels. The catheter has a dual lumen, onto the distal tip of which a balloon is welded and the proximal end of which a "Y" hub is spliced, so that guide wire Lumen forms the entrance to the central lumen for the guide wire
Features of high pressure balloon Catheter
Double cavity structure
Strong push and twist support, effectively prevent "Banana shape" balloon

Structure design
The improvement of high strength withstand voltage reduce deformation, with flexibility and push ability.

High pressure balloon
Nominal pressure reaches 10atm, the maximum rated burst pressure is reaches 22atm, low compliance.
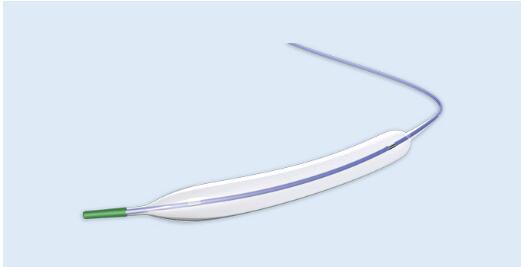
Non-compliant dilation
Maintain greater dilation at the lesion, avoiding “dog bone” effect.

Multi-specification available
Inherit PTA Balloon Catheter design; the improvement of high strength withstand voltage; Multi-specification is available.

Excellent Hydrophilic coating
Enhanced Pushability&Lesion Pressure
Enhanced Average Burst pressure
Atraumatic Tapered soft Tip
Main product of Barty Medical:
Catheter sheath
Angiography catheter
Guide catheter
PTA BALLOON CATHETER
high pressure balloon Catheter
PTCA balloon
HP PTCA balloon
System Certification
Barty has passed ISO 13485 quality management system certification approved by TUV-SUD in October, 2016; passed assessment of Chinese medical device registration quality management system in January, 2017; passed ISO 13485 quality management system certification approved by BS in July, 2017.
Hot Tags: high pressure balloon catheter, China, suppliers, manufacturers, factory, customized, OEM















